
Targets for glycaemic control in type 2 diabetes
INTRODUCTION
Increasingly, day-to-day management of people with type 2 diabetes falls to general practice nurses (GPNs).1 It is therefore important that they are familiar with the latest guidelines, and understand how to apply the recommendations to their individual patients. This includes recognising how to determine appropriate targets (for example, for blood glucose control) in each individual case.
This module will help improve awareness of the role of adequate blood glucose control in the prevention of the complications of diabetes
LEARNING OBJECTIVES
On completion of this module, you will be aware of:
- Current targets as recommended in the recent national and international guidelines
- The importance of agreeing targets with individual patients
- When to intensify drug treatment to achieve targets
- When to consider relaxing the target (on a case-by-case basis)
- The importance of good blood glucose control for the prevention of complications.
This module is one of a series of five. Others in the series are:
- Tailoring therapy to the individual patient
- Short-term complications of type 2 diabetes
- Renal considerations in patients with type 2 diabetes
- Cardiovascular considerations in patients with type 2 diabetes
This resource is provided at an intermediate level. Read the article and answer the self-assessment questions, and reflect on what you have learned.
Complete the resource to obtain a certificate to include in your revalidation portfolio. You should record the time spent on this resource in your CPD log.
Reference
1. Royal College of Nursing. Education, prevention and the role of the nurse. (Updated November 2020). https://www.rcn.org.uk/clinical-topics/diabetes/education-prevention-and-the-role-of-the-nurse
Contents
Targets for glycaemic control in type 2 diabetes
Setting targets
The focus on strict numerical targets for glycaemic control has changed in recent years, allowing greater flexibility in recognition of the needs of individuals.2
Reducing HbA1c levels is associated with a reduction in microvascular and macrovascular complications in patients with type 2 diabetes.3 Although several studies have demonstrated benefits of intensive glycaemic control in terms of cardiovascular risk and other outcomes, there is also some evidence that very low targets (46 mmol/mol, 6.4%) may be associated with increased mortality.3
There is consensus between national (National Institute for Health and Care Excellence [NICE] and the Scottish Intercollegiate Guidelines Network [SIGN]) and international guidelines (American Diabetes Association/European Association for the Study of Diabetes [ADA/EASD]) that a ‘reasonable’ HbA1c target for most adult patients with type 2 diabetes would be 53 mmol/mol (7.0%).2-4
NICE defines good glycaemic control as an HbA1c of 53 mmol/mol (7.0%), but stresses the importance of tailoring targets to the individual, to somewhere between 48 mmol/mol (6.5%) and 53 mmol/mol (7.0%).4
SIGN also recommends a target of 53 mmol/mol as reasonable to reduce the risk of complications, but suggests that a more stringent target (48 mmol/mol [6.5%]) may be appropriate at diagnosis.3 It is important to balance benefits with potential harms, including the risk of hypoglycaemia and weight gain,3 and to involve patients in decisions about targets.4
The ADA/EASD consensus statement also recommends setting individualised glycaemic targets based on patient preferences and goals, the risk of adverse events (hypoglycaemia and weight gain), and patient characteristics, including frailty and comorbid conditions.2
NICE4 recommends that clinicians should consider relaxing the target HbA1c level, on a case-by-case basis for people with type 2 diabetes who are:
- Older
- Frail
- Unlikely to achieve longer-term risk-reduction benefits, for example people with a reduced life expectancy
- At increased risk of the consequences of hypoglycaemia as a result of tight blood glucose control, e.g.
- People who are at risk of falling
- People who have impaired awareness of hypoglycaemia, and
- People who drive or operate machinery as part of their job
- Suffering from significant comorbidities, for example, for whom intensive management would be inappropriate.
Achieving and maintaining control
Patients should be encouraged to achieve and maintain agreed targets unless any adverse effects, including hypoglycaemia, or their efforts to achieve their target, impair their quality of life.4
NICE suggests that the target for HbA1c for individuals whose type 2 diabetes is managed by lifestyle and diet, or by lifestyle and diet combined with one drug that does not have an associated risk of hypoglycaemia is 48mmol/mol. For those who are treated with a drug associated with hypoglycaemia, the higher target (53 mmol/mol [7.0%]) is recommended.4 If HbA1c levels are not adequately controlled by a single drug, and rise to 58 mmol/mol (7.5%) or higher, the addition of a second glucose lowering drug is recommended, alongside reinforcing advice on diet, lifestyle and adherence to drug treatment. These patients should be supported to aim for an HbA1c level of 53 mmol/mol (7.0%).4
Initial drug therapy
Metformin remains the first line oral treatment choice in all three major guidelines, together with comprehensive lifestyle measures including weight management and physical activity.2-4
NICE recommends that metformin should be the first line oral hypoglycaemic agent unless contraindicated or not tolerated. The current guideline reminds clinicians that gradual titration of metformin is likely to improve tolerability, and that if standard release metformin is not tolerated, even when introduced at a low dose, modified release metformin may be more readily tolerated.4
Alternatives to metformin (if metformin is contraindicated or not tolerated) would include a dipeptidyl-peptidase-4 (DPP-4) inhibitor, pioglitazone*, a sulfonylurea, or a sodium-glucose co-transporter-2 (SGLT-2) inhibitor.4
*Pioglitazone is associated with an increased risk of heart failure, bladder cancer and bone fracture, and risk factors for these conditions – including increased age – should be carefully evaluated before treatment.4
First and second intensification
There is evidence for the role of clinical characteristics in informing the choice of first intensification medication (treatment with 2 non-insulin blood glucose lowering therapies in combination: dual therapy) after failure to control blood glucose with metformin and lifestyle interventions. These include the presence of established atherosclerotic cardiovascular disease (ASCVD), for which there is now evidence to support the use of SGLT-2 inhibitors and glucagon-like peptide-1 receptor agonists (GLP-1 RAs). There is also evidence that other comorbidities such as heart failure or chronic kidney disease, age and frailty, and the safety and tolerability of medication are also important when selecting first intensification medication. NICE recommends that all patients with established ASCVD or heart failure should be offered an SGLT2 inhibitor (with proven cardiovascular benefit) in addition to metformin, and that this class of drug should be considered in patients at high risk of developing CVD. SGT2 inhibitors can also be offered as monotherapy where metformin is not tolerated or contraindicated.4
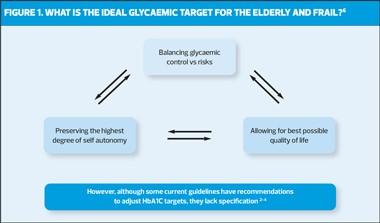
Frail and/or elderly patients
There is growing recognition that intensive glucose-lowering treatment in type 2 diabetes has limited benefits and may even be dangerous in older people.5 This should prompt clinicians to modify HbA1c targets in patients who are very frail or have limited life expectancy. Guidance developed by the University of Exeter Medical School in collaboration with NHS England recommends a target of 64 mmol/mol (8.0%) for adults with moderate frailty, and 70 mmol/mol (8.5%) for the very frail, elderly patient. The guidance warns that metformin may be contraindicated in up to 50% of frail or very frail elderly patients who are likely to have a degree of renal impairment.5 Other agents, including sulfonylureas, pioglitazone, GLP-1 agonists and SGLT2 inhibitors may all have disadvantages in frail, elderly patients. DPP-4 inhibitors have been used in frail and elderly patients, and have similar efficacy in older patients to that in younger populations.5
Frailty is fast becoming a critical complication for healthcare professionals to consider, when managing older people and their type 2 diabetes, providing it has actually been identified for the individual patient. Now, more than ever, there needs to be clear guidance and support for healthcare professionals in managing this group, especially as the numbers will continue to grow as the population ages.
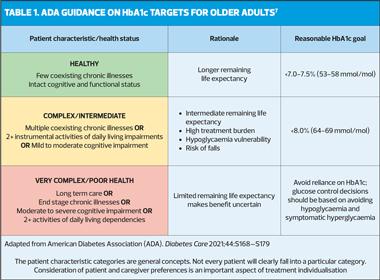
Considerations for setting blood glucose targets in elderly and/or frail patients are shown in Figure 1, and current recommendations from the American Diabetes Association (ADA) are set out in Table 1. Table 2 summarises suggested prescribing interventions for patients according to their frailty status.

In the self-assessment that follows, hypothetical case scenarios based on fictitious patients with type 2 diabetes are presented. Please refer to the relevant Summary of Product Characteristics before prescribing any of the medications mentioned.
References
1. Royal College of Nursing. Education, prevention and the role of the nurse. (Updated November 2020). https://www.rcn.org.uk/clinical-topics/diabetes/education-prevention-and-the-role-of-the-nurse [Accessed June 2022]
2. Davies MJ, D’Alessio DA, Fradkin F, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2018;41:2669-2701
3. Scottish Intercollegiate Guidelines Network. SIGN 154. Pharmacological management of type 2 diabetes; November 2017. https://www.sign.ac.uk/media/1090/sign154.pdf [Accessed June 2022]
4. NICE NG28. Type 2 diabetes in adults: management, December 2015 (Updated 2022). https://www.nice.org.uk/guidance/ng28 [Accessed June 2022]
5. Strain WD, Hope SV, Green A, et al. Type 2 diabetes mellitus in older people: a brief statement of key principles of modern day management including the assessment of frailty. A national collaborative stakeholder initiative. Diabetic Medicine 2018; 35(7):838-245
6. Abdelhafiz A, Koay L, Sinclair A, et al. The need to individualise treatment in frail older people in order to minimise the risk of hypoglycaemia and adverse outcomes. J Diabetes Nurs 2016;20:330-331. https://diabetesonthenet.com/Journal-diabetes-nursing/frailty-and-hypoglycaemia-in-older-people-with-type-2-diabetes-therapeutic-implications/ [Acessed June 2022]
7. American Diabetes Association. Older Adults: Standards of Medical Care in Diabetes–2019. Diabetes Care 2019;42(Suppl 1): S139-S147
8. Klein S, Sheard NF, Oi-Sunyer X, et al. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: rationale and strategies. Diabetes Care 2004;27(8):2067-73
9. Diabetes Education and Self Management for Ongoing and Diagnosed (DESMOND). What is the DESMOND Programme? https://www.desmond.nhs.uk/about-us [Accessed June 2022]
10. X-PERT Health. Programmes. https://www.xperthealth.org.uk/Programmes [Accessed June 2022]
11. NICE CG76. Medicines adherence: involving patients in decisions about prescribed medicines and supporting adherence; January 2009. https://www.nice.org.uk/guidance/cg76 [Accessed June 2022]
12. Boehringer Ingelheim Limited. Summary of Product Characteristics: Trajenta (linagliptin) 5 mg film-coated tablets.
13. Boehringer Ingelheim Limited. Summary of Product Characteristics: Jardiance 10 mg and 25 mg film-coated tablets.
Related modules
View all Modules