Heart failure: a roadmap for diagnosis and management in primary care
Professor Ahmet Fuat MBChB, PhD, FRCGP, FRCP, FRCPE, FESC, FPCCS, PGDiP Cardiology, CertMedEd, DRCOG, DFFP. GPSI Cardiology, Orchard Court Surgery, Darlington, County Durham
Practice Nurse 2026;56(2):16-21
In September 2025, NICE updated its guidance on the diagnosis and management of heart failure, emphasising earlier initiation and escalation of therapies and a greater role for clinicians in primary care: this article aims to help general practice nurses embed the guidance into routine practice
CONTEXT AND RATIONALE FOR THE UPDATE
The prevalence of diagnosed heart failure in England is estimated at 920,000, of whom about 63% are thought to have heart failure with reduced ejection fraction (HFrEF).1 Despite prior guideline efforts, current outcomes remain suboptimal, with frequent hospitalisations, as a result of late diagnosis, underuse of therapy, and incomplete uptitration.2
NICE estimates that earlier therapy could prevent around 3,000 deaths and 5,500 hospital admissions per year.1 The update is thus intended to ‘keep pace with changes in the treatment landscape, widen access to effective treatments, and reduce emergency hospital admissions.’1
Key changes include:
- Allowance for earlier initiation of SGLT2 inhibitors (empagliflozin and dapagliflozin) without having fully uptitrated ACE inhibitors/angiotensin receptor blockers (ARB)/angiotensin receptor-neprilysin inhibitor (ARNI) or beta-blocker first.1
- Removal of the requirement for GPs to seek specialist advice to initiate SGLT2 inhibitors.1
- Expansion of therapeutic options for heart failure with mildly reduced ejection fraction (HFmrEF) and heart failure with preserved ejection fraction (HFpEF) – mineralocorticoid receptor antagonist (MRA), SGLT2 inhibitors.1
- Increased emphasis on collaboration between primary and specialist care, multidisciplinary team working, and integration of pharmacists.1
- More robust guidance on monitoring, renal function, electrolytes, iron deficiency, and shared decision-making.1
From a primary care perspective, the shift is from passive ‘reacting’ to heart failure toward a proactive prevent, predict, suspect, test, detect, treat early (protect and perfect), monitor, and de-escalate if necessary’ paradigm.
ROADMAP FOR THE NEW PARADIGM
Below is a detailed strategy for embedding the 2025 NICE recommendations into primary care, using the following structure.
PREVENT
Goal
In high-risk patients, manage modifiable cardiovascular and kidney risk factors to prevent or delay onset of heart failure (HF).
Risk factors to target: Hypertension (HTN), diabetes mellitus (DM), atrial fibrillation (AF), coronary heart disease (CHD), lipid disorders, chronic kidney disease (CKD), obesity.
Rationale and evidence
- Hypertension is one of the strongest modifiable risk factors for future HF. A large meta-analysis (20 million participants) showed a graded association between blood pressure and incident HF.3
- Diabetes and obesity (via insulin resistance, myocardial lipotoxicity, microvascular dysfunction) are implicated in HFpEF phenotypes.
- Atrial fibrillation (AF), coronary heart disease (CHD), and chronic kidney disease (CKD) are common comorbid drivers of cardiovascular remodelling and neurohormonal activation.
- The concept of HF prevention has been elaborated by Taylor and colleagues: in their ‘Predicting and Preventing Heart Failure’ article, they argue that guidelines should better embed prevention frameworks.4
- In ‘Diagnosis and initial management of heart failure’, Fuat et al emphasize the role of natriuretic peptide testing in individuals at high risk to detect early dysfunction.5
How (implementation for GPs and general practice nurses)
- MECC (Making Every Contact Count): Train all staff (GPs, nurses, healthcare assistants [HCAs]) to opportunistically check BP, pulse, weight, BMI, waist circumference, and assess risk factors at every visit (especially in patients with hypertension, CHD, diabetes, CKD, or AF).
- Annual NHS Health Checks: Ensure eligible patients (age 40–74) attend and optimise risk factor control (BP, lipids, HbA1c, weight).
- Treatment to guideline-based targets: e.g., in hypertension, treat to < 130/80 mmHg (or guideline-specific target), in diabetes to individualised HbA1c target, and lipid to national targets.
- CVD risk review clinics: Incorporate HF risk as part of cardiovascular risk assessments in CHD/AF patients.
- Obesity management: Use referral to weight-management services, lifestyle programs, or pharmacotherapy where appropriate.
- CKD monitoring: In patients with declining eGFR or albuminuria, tighter BP and glycaemic control, avoidance of nephrotoxins.
- AF management: Rate/rhythm control, anticoagulation, and rhythm interventions (e.g. ablation) where appropriate.
By actively managing these comorbidities, many cases of HF may be delayed or prevented.
PREDICT
Goal
During long-term condition (LTC) reviews (e.g. hypertension, diabetes, CKD, AF, CHD, stroke/transient ischaemic attack [TIA], chronic obstructive pulmonary disease [COPD]), actively ask targeted questions to identify early HF symptoms and signs.
Rationale and evidence
Many patients with early HF remain undiagnosed for months or years because symptoms are subtle or attributed to comorbidities (e.g., COPD, obesity). Proactive symptom screening increases early detection. In their qualitative work, Fuat and colleagues highlight that delays often relate to low awareness and fragmentation of roles.6
The BMJ article ‘Diagnosis and Initial Management of Heart Failure,’ argues for raising awareness and earlier testing in at-risk populations.7
How
At each LTC review, use a brief HF symptom checklist:
- ·Ask about oedema (ankle swelling, abdominal bloating)
- ·Dyspnoea on exertion, orthopnoea (needing extra pillows), paroxysmal nocturnal dyspnoea (PND)
- ·Fatigue, reduced exercise tolerance, unplanned weight gain
- ·Nocturnal cough or wheeze, waking breathlessness
If any positive answers, have a low threshold to order NT‑proBNP (or refer for assessment).
- Use structured templates in the clinical record (LTC templates) that flag these questions.
- Use prompts or recall lists for high-risk patients (HTN, CHD, AF, DM, CKD, previous stroke/TIA, COPD, obesity).
- Include in nurse-led reviews or community team reviews (e.g. annual reviews by GPN).
- Document responses and escalate for testing or referral as needed.
SUSPECT
Goal
Maintain clinical vigilance: recognise symptoms and signs of HF, especially in patients with predisposing conditions, even if presentation is subtle.
Rationale and evidence
HF is predominantly a clinical diagnosis made on signs and symptoms; the key is not to delay suspicion. In early or decompensated HF, signs such as elevated jugular venous pressure (JVP), S3 gallop, displaced apex, crepitations, hepatomegaly or peripheral oedema may be subtle and depend on careful examination.
Inconsistent diagnostic pathways, limited access to tests, and unclear professional roles deter prompt diagnosis.8 In primary care, diagnostic delay remains common.7
Pumping Marvellous, a patient heart failure charity, recommends the BEAT-HF programme to raise awareness of heart failure symptoms and signs in clinicians and patients, and BEAT-TO-TREAT programme to diagnose HF more promptly in primary care settings.
How (awareness and education)
- Patient awareness campaigns: Posters, leaflets in waiting rooms highlighting red flags such as shortness of breath, swelling, fatigue, and/or waking at night breathless
- Clinician education sessions: Use case-based learning, GP/practice nurse meetings, local cardiology liaison sessions.
- Media and campaigns: Collaborate with local health networks or PCNs/ICBs to run HF awareness campaigns, e.g., ‘Check your legs, check your breath’.
- Inclusion in care pathways: Ensure HF suspicion is a recognized trigger in local chronic disease pathways, e.g., in CKD, diabetes, AF.
- Electronic prompts: In electronic health records (EHRs), flag patients with multiple risk factors for HF who have not yet been assessed.
TEST
Goal
Order the appropriate baseline investigations (NT‑proBNP, bloods, ECG, possibly chest X‑ray) in suspected HF.
Rationale and evidence
NT-proBNP (or BNP) is a sensitive biomarker to rule out HF in many patients. NICE states that a low natriuretic peptide makes HF less likely.1
ECG and basic labs (renal function, electrolytes, full blood count, TSH, liver function, iron studies) help exclude conditions that mimic HF and guide therapy.
A chest X-ray may reveal cardiomegaly or pulmonary congestion.
In community settings, primary care availability of NT-proBNP is crucial to reduce delays.
How (Implementation)
- Make NT‑proBNP available in GP practices: Negotiate with PCNs/ICBs or pathology providers to allow direct access/ordering from primary care.
- Ensure baseline blood panel kits include renal function, electrolytes, TSH, FBC, liver enzymes, iron studies (ferritin, TSAT, Hb).
- Ensure ECG availability in practice or via community ECG services.
- Initiate chest X-ray referral pathways through community imaging or local radiology.
- Implement standard protocol/order sets: Create a ‘suspected HF panel’ order set in the EHR.
- Address turnaround times: Liaise with labs to ensure prompt results (ideally < 48 h).
- Flag elevated natriuretic peptide results so that echo referral is triggered promptly.
DETECT
Goal
Refer for echocardiography (and specialist HF clinic) when patient presents with symptoms/signs plus elevated natriuretic peptide, consistent with NICE or local thresholds; maintain and validate a HF disease register to identify missing cases.
Rationale and guidance
NICE specifies that if NT-proBNP is raised (>400ng/L) and the patient has symptoms or signs of HF, they should be referred for echocardiography within 6 weeks (within 2 weeks if >2000ng/L) (ideally via specialist-access HF clinic or open access echocardiograpy) for structural and functional confirmation.1
Echocardiograms must be interpreted by specialists, with advice on diagnosis, management and coding.1
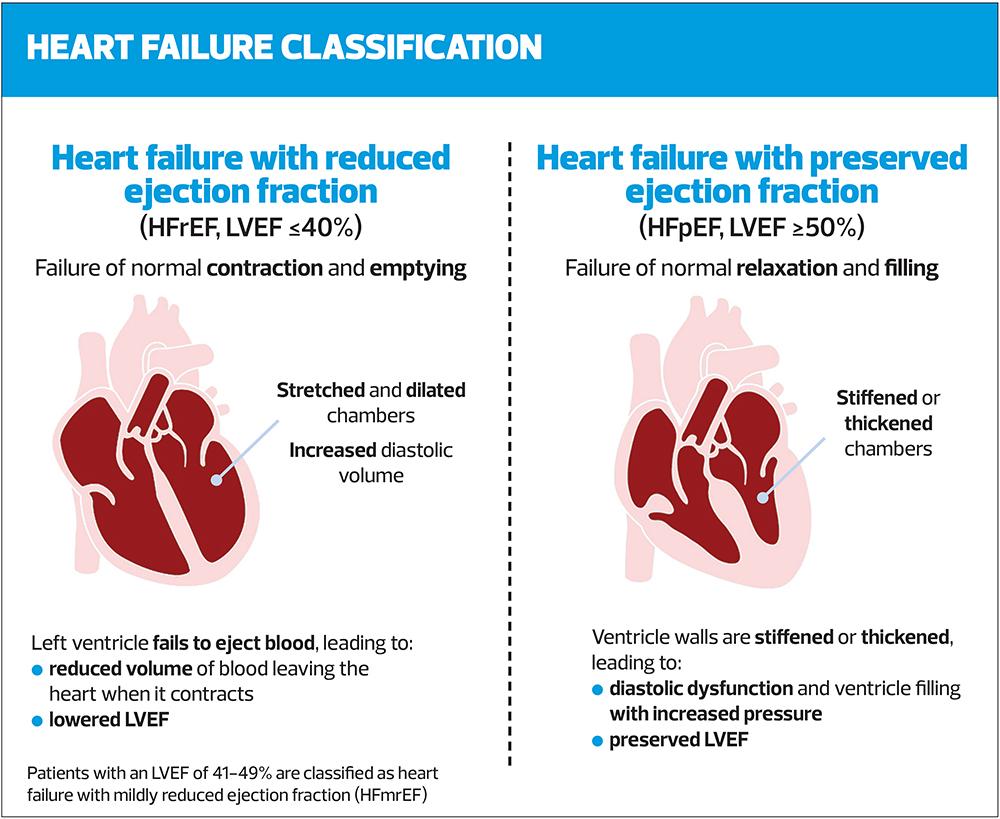
HF phenotyping:
- HFrEF: Left ventricular ejection fraction (LVEF) ≤ 40%
- HFmrEF: LVEF 41–49%
- HFpEF: LVEF > 50% +Symptoms + signs + raised NT‑proBNP + evidence of structural heart disease (left ventricular hypertrophy [LVH], left atrial enlargement [LAE]) or diastolic dysfunction).1
Regular validation of your HF disease register helps to find ‘missed’ patients (those already diagnosed but uncoded, or who meet criteria but are not on register).
How
- Open-access echocardiography pathways: Advocate locally for access to ‘fast echo for suspected HF’ clinics, or direct to HF service.
- Referral criteria in GP templates: Automatically prompt internal referral when natriuretic peptide > threshold + symptoms.
- Disease register validation audits: Quarterly or half-yearly audits comparing lists of patients with HFrEF-coded, echocardiography-identified, or natriuretic-peptide elevated, to find inconsistencies.
- Coding and specialist review feedback loops: Ask that all echocardiograms include a report with a diagnosis summary/coding suggestion and recommended follow-up.
- Patient information and letters: Ensure patients referred receive understandable leaflets about HF, the echo process, and next steps.
CODE
Goal
Apply accurate coding in the electronic medical record to enable proper care, audits, recall, and tracking.
Rationale and evidence
Accurate coding is essential to:
- Allow systematic recall and audit
- Enable performance metrics
- Avoid diagnostic ambiguity
- Ensure continuity and clarity across care settings
Under the 2025 revision, NICE expects:
- Use a broad heart failure (umbrella) code (84114007 previously G580.)
- Subcategory codes for HFREF (703272007), HFmrEF (788956000) , HFpEF (or ‘HFNEF’ in SystmOne)(446221000)
- Code for abnormal echocardiogram – echocardiogram shows left ventricular systolic (407596008) or diastolicdysfunction (407597004).
- Adequate training of primary and secondary care staff on correct codes
- Regular digital records search to pick up coding errors or uncoded cases.1
How
- Standardisation: Agree on a practice-wide coding protocol (e.g. Read code / SNOMED equivalents).
- Training sessions: Educate GPs, nurses, admin staff, and coders regarding correct coding (including differentiation among HF types).
- Automatic prompts: Create digital searches that flag patients with echo reports suggestive of HF but no HF code, or vice versa.
- Coding feedback loops: When you get echo or specialist reports, review whether the code inserted corresponds.
- Use of structured letters: Request cardiologists to supply suggested codes in outpatient letters.
- Audit and reconciliation: Quarterly review of the HF register, coding mismatches, and missing cases.
PROTECT
Goal
In patients with confirmed HF (especially HFrEF) or symptomatic HFmrEF, institute evidence‑based therapies and lifestyle measures to prevent deterioration. HfPEF should also be managed appropriately to prevent hospitalisations and improve quality of life.
Rationale and pharmacotherapy
In HFrEF, the guideline retains the concept of four foundational therapies (the ‘four pillars’): ACEI (or ARNI), beta-blocker, MRA, and SGLT2 inhibitor. However, the 2025 update allows greater flexibility: therapies may be initiated earlier, concurrently, and without requiring full uptitration of prior drugs.1
If patients remain symptomatic on maximal therapy, switching ACEI to ARNI is an option.1
For HFmrEF, consider ACEI/ARB, beta‑blockers, MRAs and SGLT2 inhibitors (per their evidence) to reduce hospitalisation.1
For HFpEF, the 2025 update permits considering MRA and SGLT2 inhibitor in symptomatic patients.1
Additionally, the guideline emphasises lifestyle measures, rehabilitation, patient education, and management of comorbidities.1
How
- Hand-held records/patient passports: Provide patients with printed (or digital) medication journey booklets identifying their 4‑pillar therapy (or less in HFmrEF and HFpEF), side effects, targets, and sick day rules.
- Education for clinicians and patients: Use in-practice teaching sessions, patient workshops, and printed leaflets.
- Digital audit/recall platform searches: Quarterly searches to identify patients not on all appropriate therapies or not uptitrated.
- Shared decision making: Incorporate patient preferences, comorbidity, frailty, and tolerability when prescribing and accelerating therapy.
- Referral back to HF specialist for complex cases: If initiation or titration becomes high risk (e.g. borderline renal function, hyperkalaemia, hypotension).
- Rehabilitation referral: Enrol patientsin cardiac rehabilitation or HF-specific rehab programmes.
- Lifestyle support: Dietary advice (salt restriction, fluid management), exercise counselling, weight control, smoking cessation, vaccinations.
PERFECT
Goal
Strive for maximal tolerated doses of evidence-based drugs, apply sick day rules, and ensure therapy is paused only when necessary, and restarted promptly.
Rationale
Under-treatment and under-titration are common reasons for suboptimal outcomes (the treatment gap). The 2025 NICE update encourages more assertive titration within safety limits, with more frequent monitoring early in initiation.1
Sick day rules are increasingly recommended for patients on ACEI/ARB/ARNI/MRA/SGLT2 inhibitors: temporarily suspend these drugs in cases of dehydration, vomiting, diarrhoea, or acute kidney injury risk.
How
- Structured titration plans: Within each patient’s hand-held record or EHR, schedule titration intervals (e.g. every 1-2 weeks) with specific target doses.
- Monitoring schedule: Renal/electrolytes 1–2 weeks after starting or escalating, then every 3–6 months.1
- Alerts and prompts: Electronic reminders or nurse recall for monitoring and dose escalation.
- Educate patients on sick day rules: Clearly explain when to temporarily stop ACEI/ARB/ARNI/MRA/SGLT2 (e.g. vomiting, diarrhoea, volume depletion, serious intercurrent illness) and how/when to restart.
- Rapid resumption: As soon as the intercurrent illness resolves, reintroduce therapy.
- Regular audits: Use digital searches to identify patients on submaximal doses, or who delayed restart after illness.
DE‑ESCALATE
Goal
In frail patients or those with limiting comorbidities, review and consider reducing or discontinuing therapies, or deactivating devices (CRT/ICD) in shared decision-making frameworks.
Rationale
Not all patients benefit from full therapy indefinitely. In those with advanced frailty, polypharmacy, or limited life expectancy, a balanced approach is needed. The guideline underscores tailoring therapy to comorbidity, prognosis, and patient preferences (frailty-aware decision making).1
In device therapy (cardiac resynchronisation therapy [CRT], implantable cardioverter-defibrillator [ICD]), deactivation may be appropriate in the context of terminal illness or futility.
How
- Frailty assessment protocol: Use validated tools (e.g. Rockwood Clinical Frailty Scale) during regular reviews.
- MDT discussion: Host quarterly or biannual MDT meetings (GP, cardiology, geriatrics, HF nurse) to review borderline patients.
- Shared decision-making discussion: With patient/caregiver, consider discontinuing or dose-reducing medications with marginal benefit or high side-effect risk.
- Device review pathways: Engage cardiology/arrhythmia teams to review CRT/ICD necessity; protocols for device deactivation should be in place locally and ethically managed.
STRENGTHS, CHALLENGES AND BARRIERS TO IMPLEMENTATION
Strengths of the 2025 update
- Earlier access to therapy: Removing rigid sequential titration means no ‘therapeutic inertia’ barrier.
- Empowering primary care: GPs/GPNs can initiate more therapies without mandatory specialist gatekeeping.
- Broader pharmacologic scope: Extending options into HFmrEF and HFpEF addresses prior gaps
- Multidisciplinary inclusion: Recognising pharmacists, specialist prescribers, and upstream diagnostics within the MDT.
- Modern monitoring guidance: More consistent recommendations for renal/electrolyte checks, iron deficiency assessment, safety parameters.
Key challenges and barriers
- Access to diagnostics in primary care
- In some areas, NT-proBNP is not directly accessible to GPs. Echocardiography is not always open-access.
- Delays in testing jeopardise rapid diagnosis.
- Resource and staff constraints
- Time pressures in consultations, especially to titrate multiple medications.
- Insufficient training or confidence in HF in primary care.
- Uptitration inertia and safety fears
- Concerns about hypotension, renal deterioration, hyperkalaemia deter dose escalation.
- Unclear transfer of responsibility between primary and specialist care.
- Coding inconsistencies and register gaps
- Mismatches between clinical diagnosis and coding lead to omissions in recall/audit.
- Lack of feedback from cardiology on coding best practices.
- Comorbidity burden and competing priorities
- Many HF patients have multimorbidity (CKD, COPD, frailty) limiting ideal therapy.
- Balancing guideline-driven therapy with individual patient context is complex.
- Patient engagement and adherence
- Polypharmacy burden, side effects, educational gaps impede uptake.
- Sick day rules and re-initiation of medication often poorly understood.
- Frailty and de-escalation complexity
- No clear thresholds or algorithms for when to de-escalate in real-world practice.
- Local variation in service configuration
- Some regions lack integrated HF clinics or care networks, impeding feedback loops.
Indeed, one qualitative study found that GPs and HF nurses reported persistent barriers over a decade: lack of diagnostic access, variable care pathways, and ambiguity about responsibility.6
Mitigation and recommendations
- Local engagement: GP practices should engage with ICBs, cardiology networks, and pathology providers to negotiate direct access to NT-proBNP and community echo pathways.
- Use of shared care protocols: Co-developed protocols for initiation and titration (shared between primary and secondary care) reduce uncertainty.
- Education and mentorship: Pair less experienced GPs with cardiology leads, heart failure nurses, or external tutors (e.g. from local trusts).
- Nurse-led titration clinics: Practice nurses or pharmacists can lead dose escalation under protocol with GP oversight.
- Audit and feedback: Use internal dashboards to highlight performance gaps (e.g. patients not on full therapy) and monitor improvement.
- Patient engagement tools: Leaflets, apps, telemonitoring, and home-monitoring kits help adherence and early detection of decompensation.
- Gradual rollout: Start with high-risk pilot groups (e.g. patients with HFrEF known and under-treated) before extending to broader cohorts.
- Governance and documentation: Clear documentation of decisions (escalate, de-escalate, titration plan) within the patient record.
CONCLUSION
In summary, the 2025 NICE update marks a paradigm shift toward more proactive, earlier, and primary-care-integrated management of heart failure.
While the updated guideline represents a step forward, there remain areas for further evolution, including greater clarity on de-escalation thresholds, tailored protocols for multimorbidity – many HF patients have CKD, COPD, anaemia, etc. and more guidance on prioritization, drug interactions, and competing management is needed.
More granular guidance is also needed on which patients require more frequent renal/electrolyte monitoring – for example, those with borderline eGFR, prior hyperkalaemia, advanced age.
The guideline update focuses on chronic HF; nonetheless, outpatient management of decompensation is gaining interest (e.g. home IV diuretics), and there is some evidence that it may reduce 30-day hospitalization and mortality.
The guideline uses fixed NT‑proBNP cutoffs; in this author’s opinion, more age- and comorbidity-adjusted thresholds might reduce inappropriate referrals.
It is also essential to ensure that under-served, rural or resource-poor practices are supported to access diagnostics, and are not left behind.
The guideline aims to address current suboptimal outcomes that result from late diagnosis, underuse of therapy, and incomplete uptitration, and general practice plays a vital role in securing these ambitions.
To succeed, primary care teams must proactively secure diagnostic access, train staff, adopt recall/audit processes, and foster strong collaboration with HF specialist services. The benefits – reduced mortality, fewer hospital admissions, improved quality of life – make this transformation essential.
References
- NICE NG106. Chronic heart failure in adults: diagnosis and management; updated 2025. https://www.nice.org.uk/guidance/ng106
- Howlett J, Comin‑Colet J, Dickstein K, et al. Clinical practices and attitudes regarding the diagnosis and management of heart failure: findings from the CORE Needs Assessment Survey. ESC Heart Failure. 2017;5(1):172-183 dot:10.1002/ehf2.12205.
- Buffer PK, Jahangiry L, Jain S, et al. Blood pressure, hypertension and the risk of heart failure: a systematic review and meta-analysis of cohort studies. Eur J Preventative Cardiology 2024; 31(5): 529-556 https://doi.org/10.1093/eurjpc/zwad344
- Taylor CJ, Beezer J, Fuat A, et al. Predicting and Preventing Heart Failure. BMJ Considerations in Medicine. 2024;3:e000007. https://considerations.bmj.com/content/3/1/e000007
- Pellicori P, Ahmed FZ, Beezer J, et al. Diagnosis and Initial Management of Heart Failure. BMJ Considerations in Medicine. 2024; 3:e000008.https://considerations.bmj.com/content/3/1/e000008
- Hancock HC, Close H, Fuat A, et al. Barriers to accurate diagnosis and effective management of heart failure have not changed in the past 10 years: A qualitative study and national survey. BMJ Open. 2014;4(3): e003866.
- Ahmed FZ, Beezer J, Fuat A, et al. Managing Heart Failure in the Longer Term. BMJ Considerations in Medicine. 2024;3:e000013 https://considerations.bmj.com/content/3/1/e000013
- Fuat A, Hungin APS, Murphy JJ: Barriers to effective diagnosis and management of heart failure in primary care.BMJ 2003; 326:196-200
- Bahar J, Rahman A, Wong G, et al. Outpatient treatment of decompensated heart failure: A systematic review and meta‑analysis. ESC Heart Fail 2025;12(2):761-769
- Charman SJ, Okwose N, Taylor CJ, et al. Feasibility of the cardiac output response to stress test in suspected heart failure patients. Fam Pract 2022;39(5):805‑812.
- Fuat A, Murphy JJ, Hungin APS. The Diagnostic Accuracy and Utility of a B‑type Natriuretic Peptide Test in a Community Population of Suspected HF. Br J Gen Pract 2006;56(526):327-33
- Cheema HA, et al. Efficacy and safety of SGLT2 inhibitors for heart failure with mildly reduced or preserved ejection fraction: systematic review and meta‑analysis. Front Cardiovasc Med 2023;10:127378. https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2023.1273781/full
Related articles
View all Articles